AUC24 Vancomycin Bayesian-based dosing: Increasing Therapeutic Target Attainment with Decreased Monitoring Costs
In this article:
Note: The new vancomycin dosing guidelines have been officially released. Learn what’s changed and how they’ll impact you by reading our guide to the new vancomycin dosing changes here.
As many health systems prepare to implement the forthcoming vancomycin dosing and monitoring guidelines, a common question is regarding what effect these guidelines will have on the frequency of obtaining vancomycin levels.
Our scientific team conducted an analysis looking at these trends recently presented at IDWeek, an international scientific conference that brings together experts in the field of infectious diseases.
Executive Summary
In this study, we compare therapeutic range attainment across five hospitals using trough-based dosing versus five hospitals using Bayesian-supported AUC24 vancomycin dosing.
We believe these results are compelling – even for hospitals that have not moved entirely to AUC based dosing and are still monitoring some patients based on vancomycin trough levels. From the cohort, the pharmacists dosing with DoseMeRx Bayesian dosing achieved significant improvements in their vancomycin dosing performance.
The Results
- Mean vancomycin dose was significantly lower in the Bayesian-based dosing cohort compared to the standard-practice cohort, suggesting a lower likelihood of acute kidney injury risk.
- The Bayesian-dosing cohort achieved a significantly higher proportion of doses within both the target trough and target AUC24 therapeutic ranges.
- Using model-informed precision dosing to target AUC24 avoided increased laboratory cost, with a median number of assays per day of therapy slightly lower than for standard-practice trough-based dosing.
- Vancomycin dosing protocols could potentially be adjusted to reduce the number of concentrations taken by approximately one-third while maintaining efficacy and safety.
Introduction
Recent literature has broad support for switching from trough-based to AUC24-dosing of vancomycin.
- Vancomycin efficacy is optimally predicted by the 24 hour area under the concentration-time profile (AUC24), yet for clinical ease, surrogate trough (Cmin) targets have traditionally been used.
- The variable relationship between trough and AUC24 has been demonstrated to lead to vancomycin overexposure in approximately 40% of patients, who will achieve a therapeutic AUC24 with a trough lower than the traditional 10-20 mg/L target, with unnecessarily increased nephrotoxicity risk (Chavada R, et al. Antimicrob. Agents and Chemo. 2017;61:e02535.)
- Traditional AUC24-based dose calculation methods involve complex analytic pharmacokinetic calculations that require both peak and trough drug levels, increasing cost and time compared to trough-based dosing.
- Recent literature (Rybak et al. ASHP draft vancomycin guidelines 2019) suggest that Bayesian dosing tools alleviate the cost and difficulty of implementing AUC24-based vancomycin dosing in order to improve patient outcomes.
While there is broad literature support for the use of Bayesian dosing tools such as DoseMeRx for implementing vancomycin AUC24-based dosing, understanding the impact of these tools in clinical practice, rather than clinical trials was the clear next research step.
Study Objective
Compare therapeutic range attainment amongst five hospitals across the US, Europe, and Australia using trough-based dosing versus five hospitals using either Bayesian-supported AUC24 dosing or Bayesian-supported trough based dosing with AUC24 prominently displayed at the time of decision-making.
Methods
- De-identified data from retrospective chart reviews for patients receiving vancomycin were available from five hospitals across the US, EU, and Australia that used a trough-based dosing method.
- De-identified data from five matched-size hospitals that implemented Bayesian-based AUC24 dosing (n=2) or trough-based dosing with AUC24 prominently presented (n=3) using commercially available software (DoseMeRx) were collected.
- The proportion of doses in the therapeutic target range was determined for each hospital, and the number and cost of therapeutic drug monitoring (TDM) levels required were compared.
Results
Patient Data
After data collection, we had a very large data set with:
- Five hospitals using clinician-led trough-based dosing without Bayesian or model-informed precision dosing had data for 409 adult patients, represented 13,775 doses, and 5,660 drug levels collected.
- Five hospitals using model-informed precision dosing (DoseMeRx) had data for 1,499 patients, representing 21,107 doses, and 7,310 drug levels collected.
Clinical Outcomes
- Mean dose was significantly lower in the model-based dosing cohort (1118 mg vs 1413 mg; p < 1×10-15).
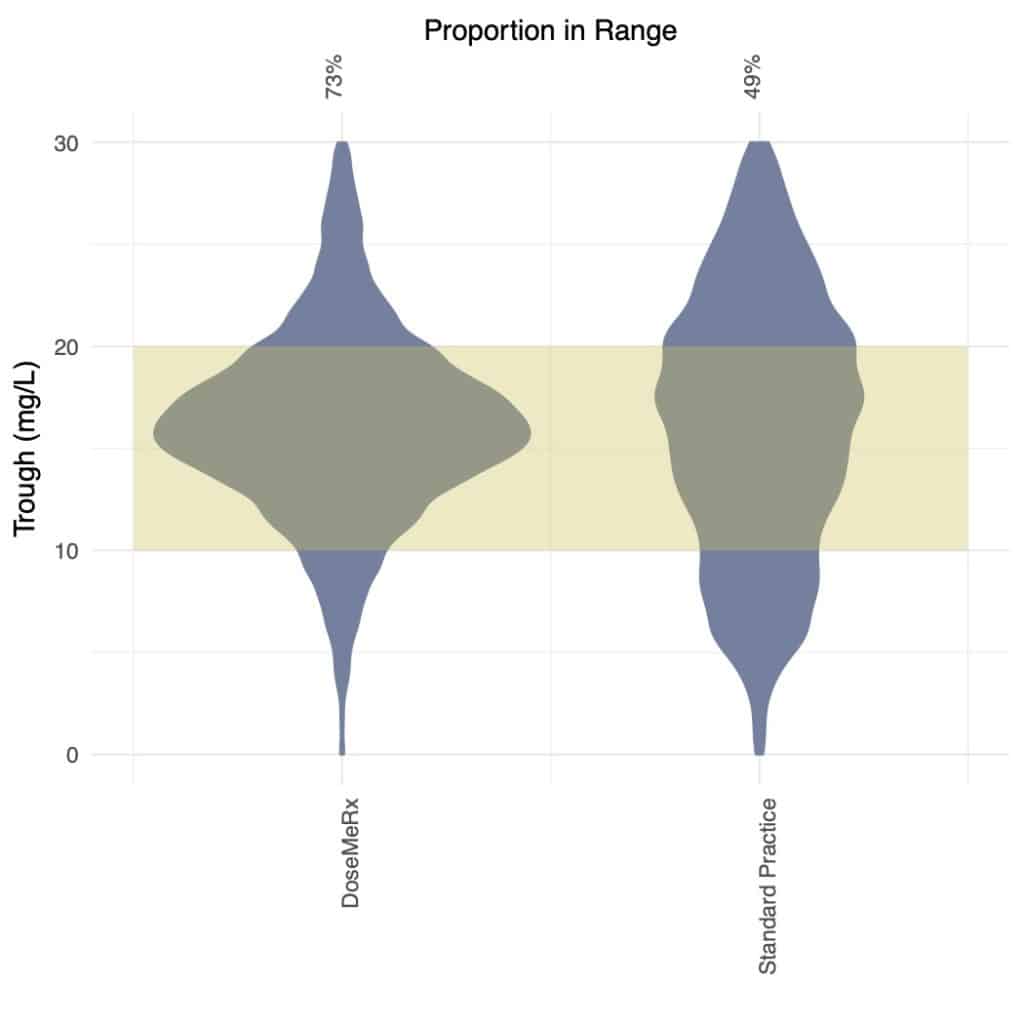
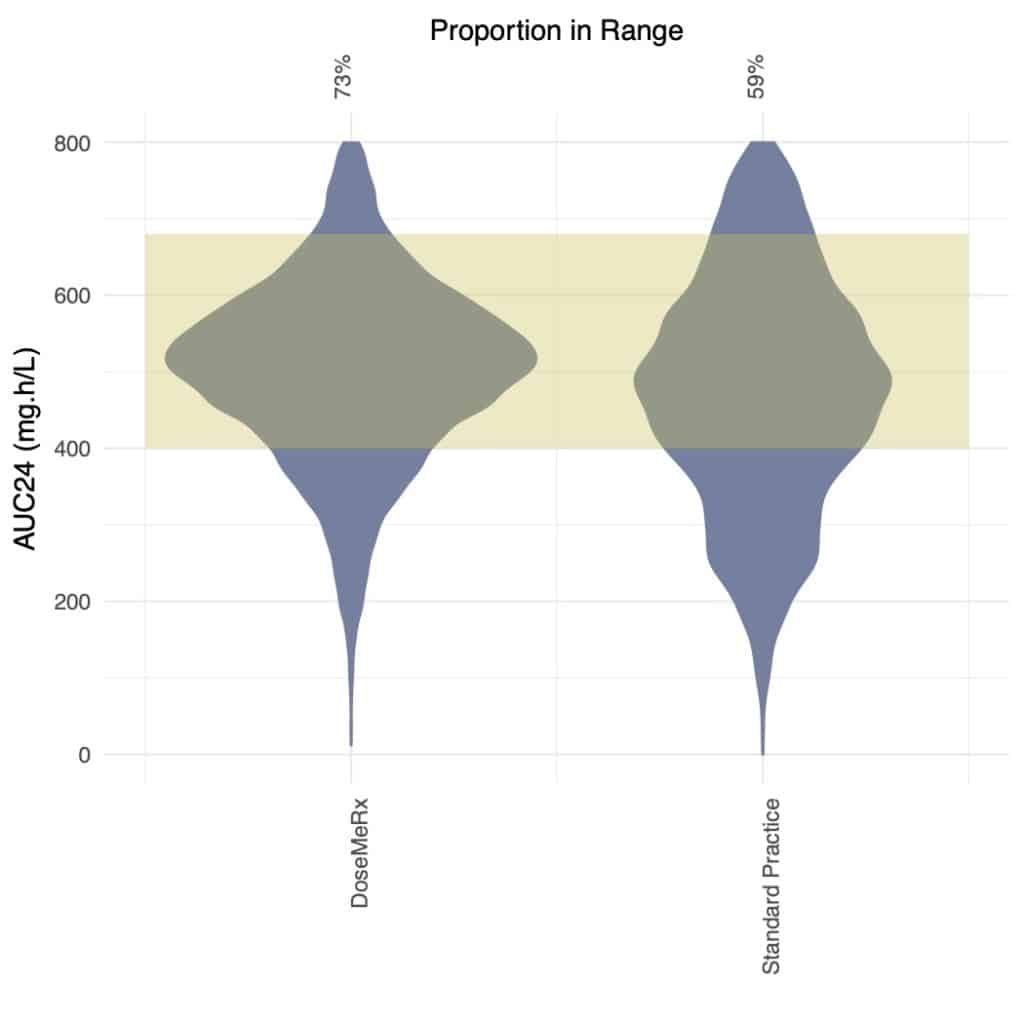
- Bayesian dosing hospitals had a significantly higher proportion of doses achieving the therapeutic target for both AUC24 (73% vs 59%; Chavada et al. therapeutic target) and trough (73% vs 49%. p < 1 x 10-15; KS test).
- Variability was significantly higher in standard practice hospitals than those using DoseMeRx for trough-based target attainment with SD=6.9 mg/L vs 5.6 mg/L; AUC24 SD=154 mg.h/L vs. 137 mg.h/L. Standard Practice D.
| Standard Practice | DoseMeRx | |
|---|---|---|
| # Patients | 409 | 1499 |
| # Male | 268 (65.5%) | 834 (55.6%) |
| Age (years) | 60.0 (18.8-96.7) | 66.7 (18.4-100.5) |
| Weight (kg) | 80.1 (44-200) | 79.9 (30-220) |
| Height (cm) | 171.0 (150-198) | 169.1 (122-202) |
| Doses Per Course | 11.7 (11.3) | 13.0 (15.3) |
| Conc. Per Course | 4.4 (4.8) | 3.5 (3.1) |
| Dose (mg) | 1413 (740) | 1118 (385) |
| Concentration (mg/L) | 19.6 (7.5) | 17.8 (6.4) |
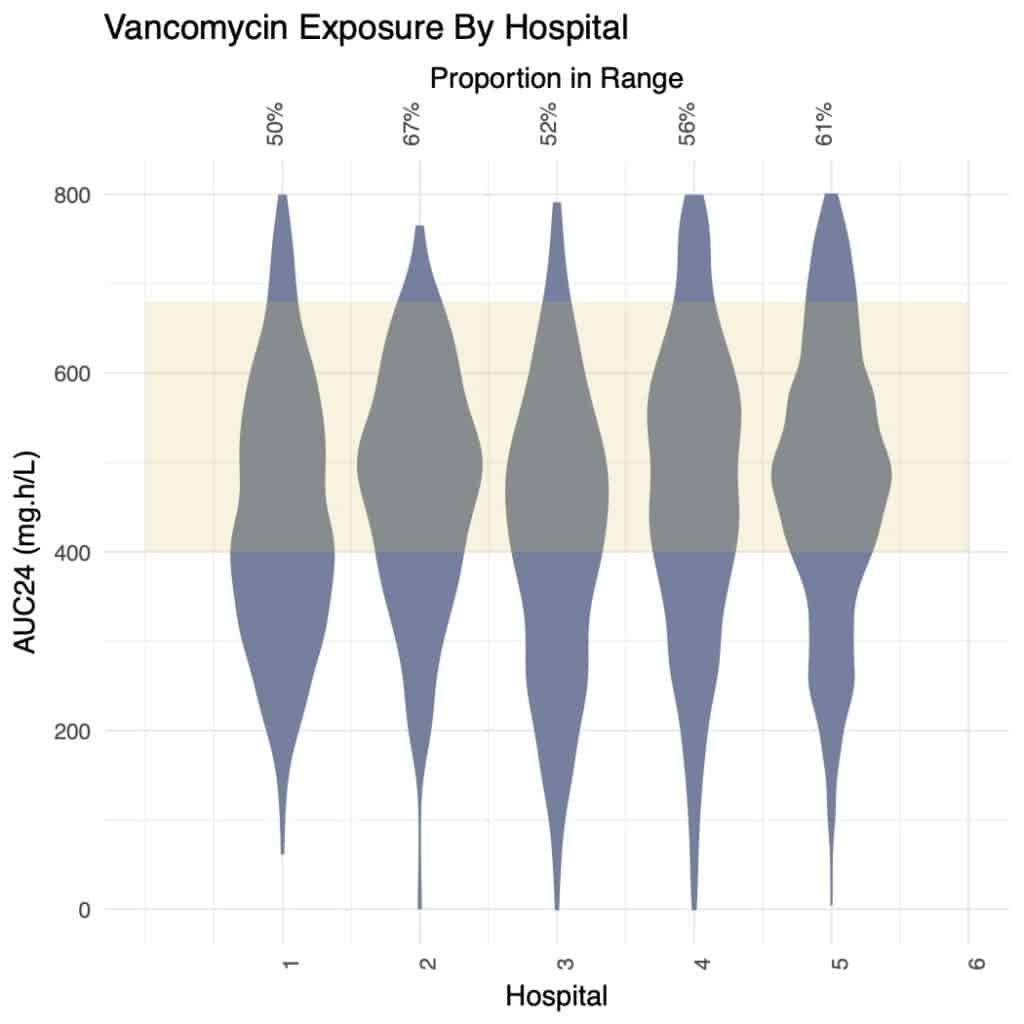
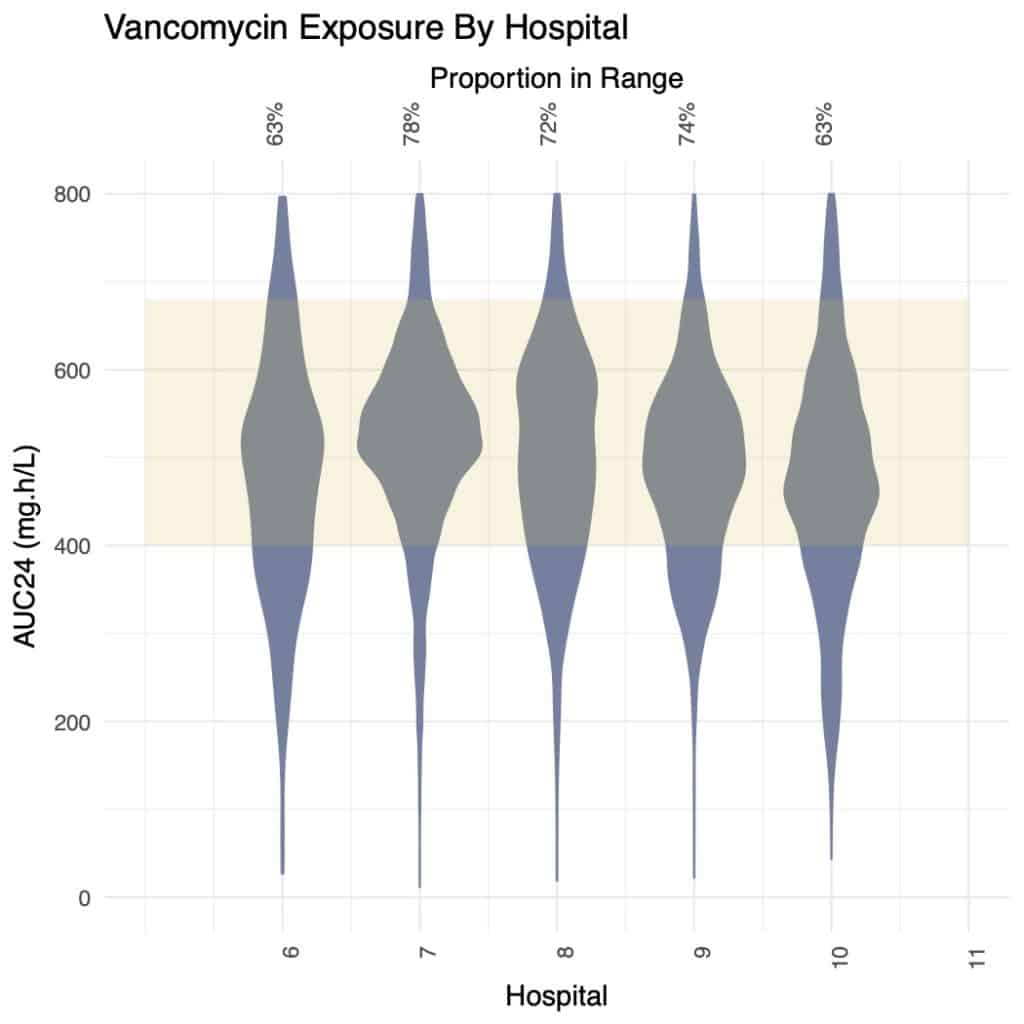
Cost Outcomes
Concentrations Per Day
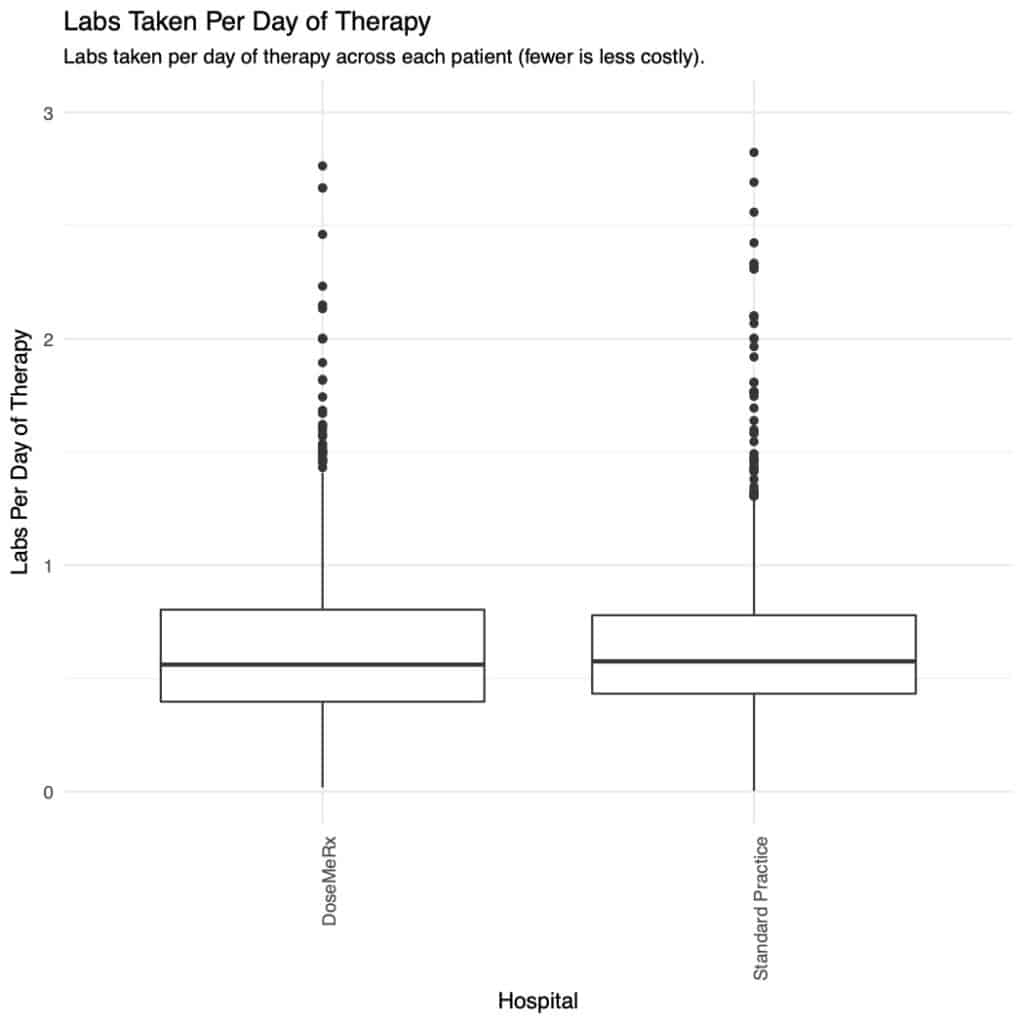
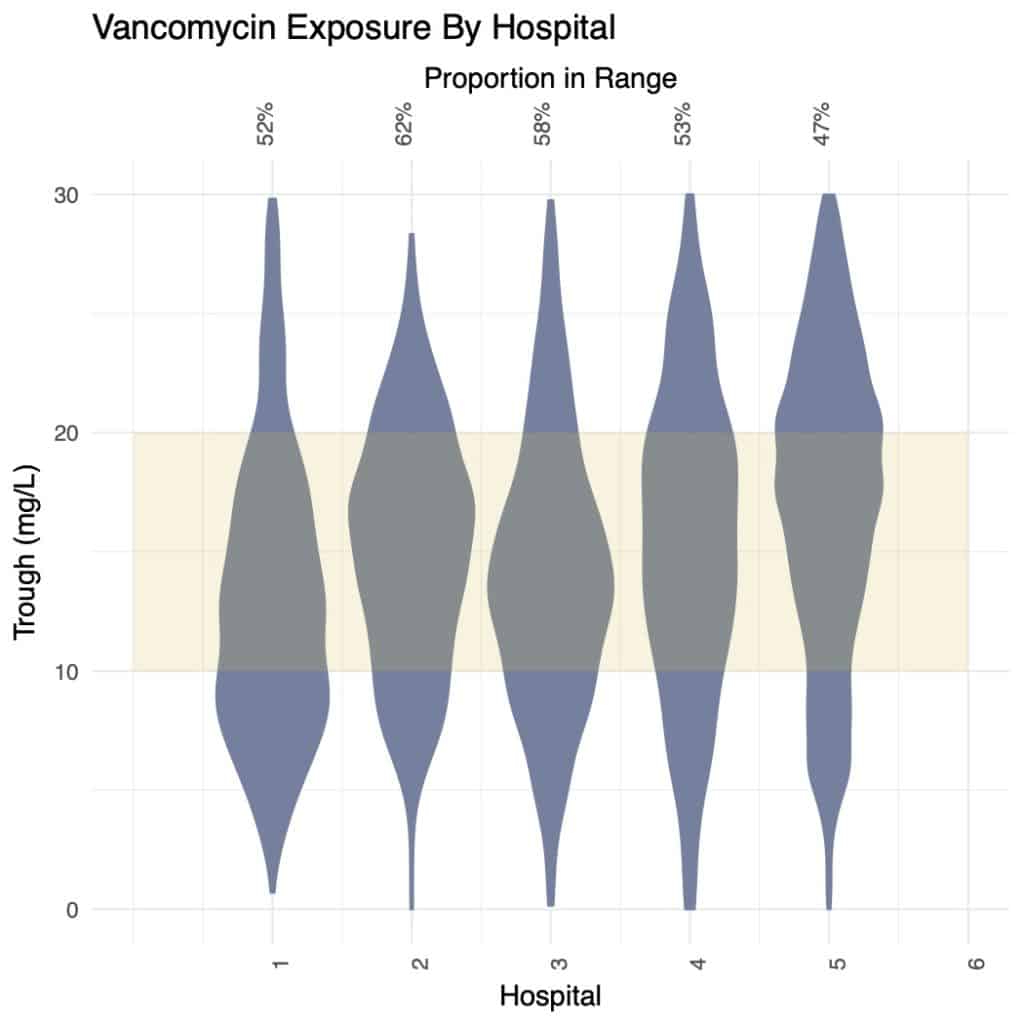
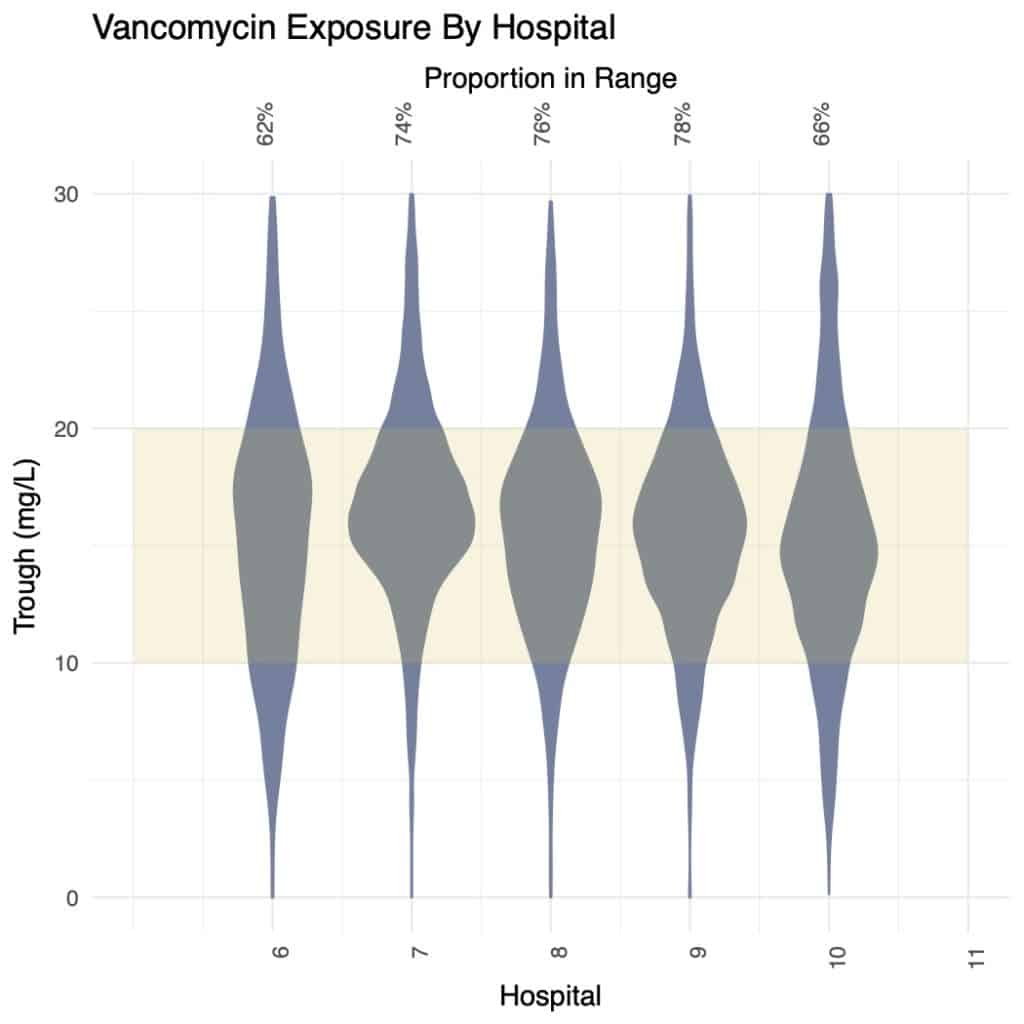
- The median number of concentrations taken per day of vancomycin therapy were significantly lower at 0.56 for the DoseMeRx and and 0.58 for the standard-practice cohorts respectively (p<0.002; KS test).
- More variance was present in the number of concentrations taken per-day between hospitals within the group than between standard-practice hospitals (1-5) and Bayesian-dosing hospitals (6-10).
- The Bayesian-dosing hospital with the fewest concentrations per day of therapy (Hospital 7; figure D) not only took fewer levels (0.36/day vs mean 0.58/day; 36.0% fewer concentrations; p<0.05), but also performed significantly better than the mean (78% vs 73% doses in AUC range), suggesting that Bayesian dosing may permit safe reduction in the number of vancomycin levels taken.
Discussion
For pharmacy teams wishing to improve their practice, these results appear quite compelling. In combination, hospitals using DoseMeRx have:
- Lower vancomycin doses (i.e. lowering the risk of AKI)
- Improved proportion of therapeutic doses (i.e. improved efficacy)
- Lower laboratory costs, and easier management (fewer drug levels)
This presents an interesting proposition – for these hospitals using DoseMeRx resulted in lower risk, and improved efficacy, and lowered cost – in combination, an outstanding result.
Download the Scientific Poster
Authors: Paul E. Sabourenkov, Mbioinf(Adv), Robert C McLeay, PhD
Originally Published ID Week Annual Meeting; October 4, 2019; Washington, DC, Poster Presentation #1599
Related Posts
Here are some more vancomycin resources for you:
